Researchers develop new eDNA tool to detect tuna (eel)
10 March 2023
As part of the national programme Lakes380 co led by Cawthron and GNS Science, researchers have developed a new environmental DNA-based tool to detect tuna (shortfin and longfin eel) in Aotearoa New Zealand’s lakes and rivers. This new tool will help to understand the presence and distribution of this taonga species in our waterways.
Tuna are found in lakes and rivers across the country. However, there has been a national decline in their populations due to habitat destruction and commercial harvest. Understanding where they live and how their populations have changed is critical to their protection.
Traditional methods of monitoring tuna, such as nets, visual surveys or electrofishing can be time consuming, costly, and limit the number of surveys that can be undertaken, as well as potentially being environmentally invasive.
To help create a more efficient way of identifying the presence of tuna, Cawthron PhD student Georgia Thomson-Laing has led a research project to develop a new environmental DNA (eDNA) method that can rapidly detect them in water and sediment samples.
“This new method will allow us to undertake surveys for tuna at a resolution and scale not previously possible,” said Thomson-Laing.
“This means we can get a much more accurate picture of where tuna populations are declining, so that we can more effectively target these environments for protection or restoration.”
The term environmental DNA refers to traces of DNA collected from environmental samples (such as soil, sediment, water, air). The sources of this DNA vary, but can include DNA shed through faeces, mucous, skin, eggs, or pollen.
Thomson-Laing is also using this method to work on lake sediment cores to detect the presence of tuna in the past, which will contribute to understanding how their populations have declined over time.
In addition, Cawthron scientists are involved in research led by Otago University’s Amandine Sabadel and funded by the Royal Society Te Apārangi to understand where tuna spawn, and how their larvae are dispersed. Adult tuna leave Aotearoa New Zealand and swim thousands of kilometres across the ocean to spawn somewhere in the western South Pacific Ocean before they die, and the larvae then travel back to grow and mature.
Researchers are using two methods to track migration; one involves identifying chemical markers specific to the local environment that could make it possible to locate an animal at a specific time or place. The second approach, being led by Cawthron molecular surveillance team leader Xavier Pochon, uses the new eDNA tool to identify the presence or absence of tuna in any given environment.
“eDNA surveillance uses genetic rather than chemical markers, but the two technologies will be integrated to try and track our eels’ migratory pathways and pinpoint their spawning location andto better understand their life-cycle and protect them ,” said Pochon.
Both of these projects are examples of Cawthron’s world-leading and innovative molecular capability to develop a range of eDNA methods. These new tools offer universal and cost-effective ways to measure biodiversity and detect the presence of organisms, including pests. It is a fast-moving and innovative field, and increasingly environmental agencies and industry are adopting it to support environmental management and species detection in a broad range of areas such as biodiversity conservation, biosecurity, agriculture, and environmental management.
The research on the new environmental DNA-based tool to detect tuna has recently been published in an international journal – you can read the full article here.
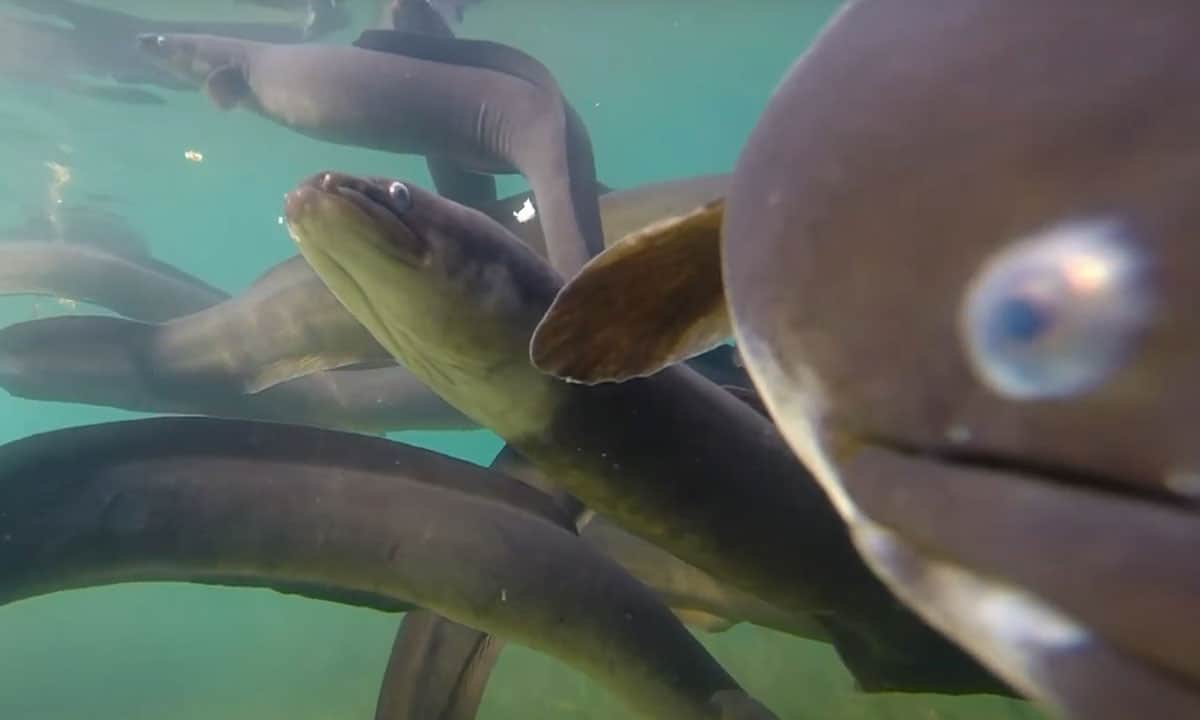
Image: Cawthron Institute. Tuna (eels) underwater.
